Understanding the Life Cycle of Internal Parasites
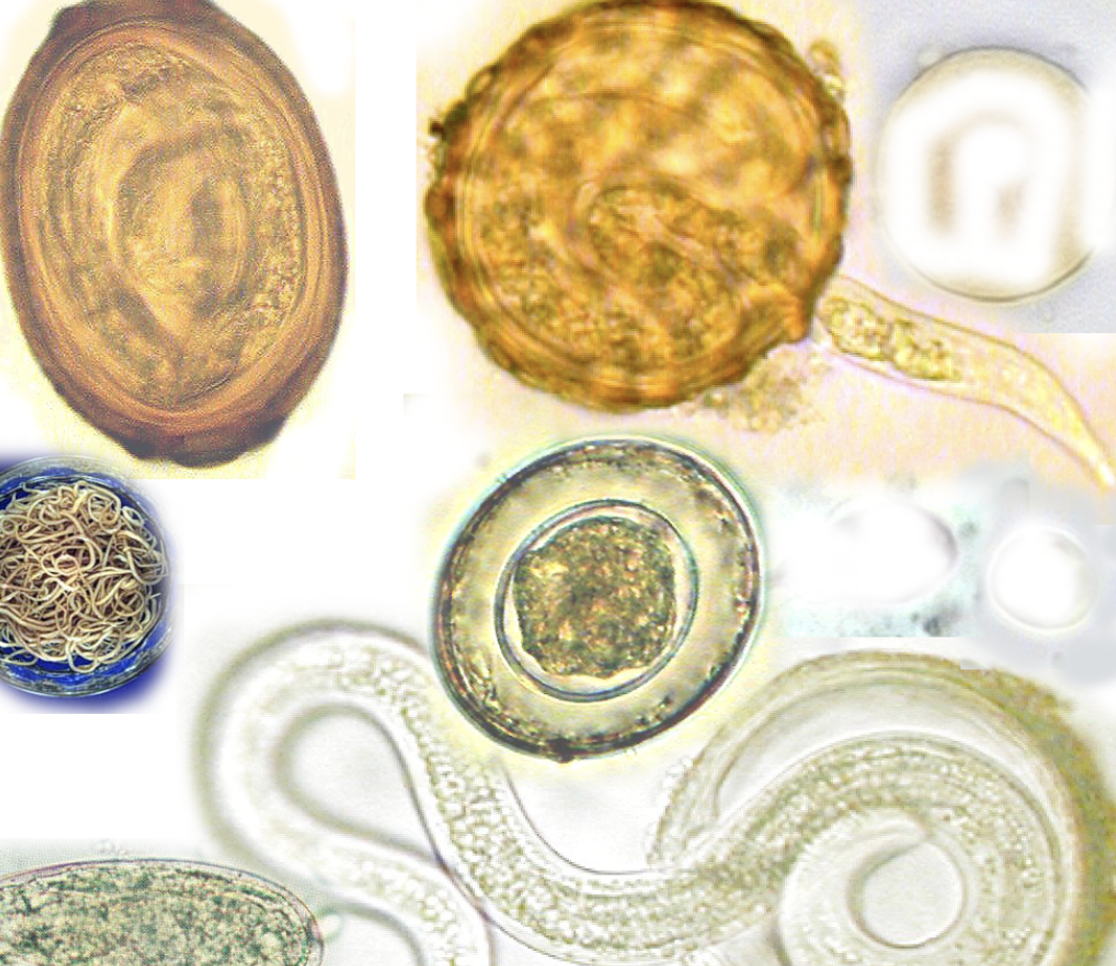 In this chapter, we’re going to discuss the life cycle of helminths and sporozoa as well as symptoms.
In this chapter, we’re going to discuss the life cycle of helminths and sporozoa as well as symptoms.
Helminth Life Cycles
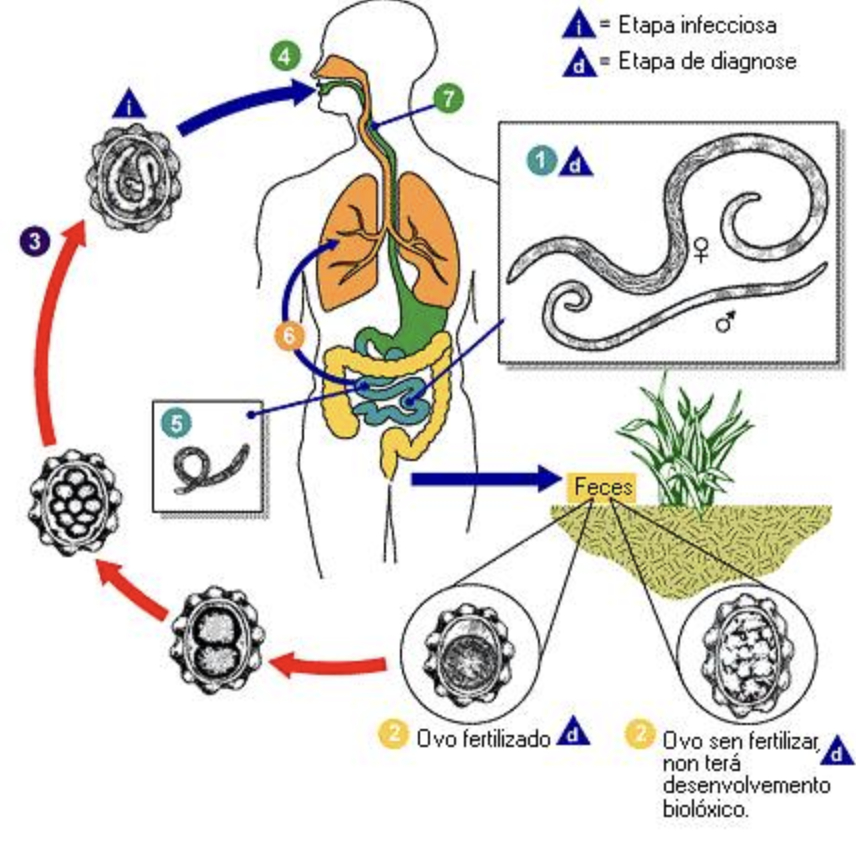
Helminths (parasitic worms) infect humans through life cycles that are remarkably complex. A single organism may pass through multiple distinct biological stages (eggs, larvae, adults), transition between soil or water and one or more living hosts, and navigate the internal geography of the human body with a specificity that continues to inform how we think about diagnosis and treatment. Infection can occur through ingestion of eggs or larvae in contaminated food or water, through direct skin penetration by larvae, or through the bite of a vector. Once inside the host, the journey typically involves intestinal maturation, migration through tissues and lungs, and eventually the release of eggs back into the environment through feces. The cycle then starts over again.Understanding the life cycle of helminths is important in terms of treatment because not all medicinal agents are able to target all of the different stages in the life cycle of a parasite. Though a particular medicine might be able to target larvae, it may not be able to target and kill eggs that are in the body. Eggs take a certain amount of time to mature which produces a cyclical symptom picture wherein an anti-parasitic medicine that targets larvae successfully reduces the “worm load” but then within days or weeks, eggs hatch and the worm load increases again. Symptoms seem to get better as larvae are killed, but then they worsen again when eggs hatch inside the body. To successfully get rid of a parasite infection, you have to consistently treat the parasites, but you also need to be aware that parasites can become resistant to certain medicinal agents if you use them too often.

Click here to schedule a health coaching call with us.
In other words, let’s say that you have a heavy worm load of hookworms. You might be anemic with brain fog, and have achiness throughout your body, a lack of appetite and gut inflammation, and other strange symptoms. If you take pyrantel pamoate for 3 days in a row according to the instructions on the bottle, you should then stop taking the pyrantel pamoate. You would not take another dose about 35 days later (3 days of dosing followed by several weeks off) because hookworms have a life cycle of 5 to 9 weeks. Your goal, as with all parasites, is to kill a batch of parasites monthly and avoid exposing the parasites to one specific medicine on a regular basis such that the parasites can develop a resistance to it.Key Stages in the Helminth Life Cycle
Transmission and Infection The entry point to the human body depends on the species. Some helminths, roundworms and tapeworms, for example, gain access through the ingestion of contaminated food or water. Others, like hookworms, bypass the digestive route entirely, with larvae penetrating intact human skin directly from contaminated soil.Many of the clients that we’ve worked with who have a heavy worm load of one or more types of helminths, become very nervous about their exposure to the environment and develop a fear of interacting with nature or with environments where parasites are transmitted easily. This makes sense in some respects, but it’s important to note that while exposure to parasites is almost ubiquitous, infection does not always occur. For example, I worked in long-term care facilities for 5 years when I was a college student. I went to a variety of long-term care facilities as an agency worker, so I was exposed to a lot of human poop, blood, and other bodily fluids during that time. I didn’t ever develop a parasite infection as a result of this work though. Why not? My guess is that I was better nourished as a young woman and less stressed at that time in my life. My body was able to resist and overcome parasite infections at that time in my life.
My family and I became host to helminths when Lydian became a teenager. Based on what I know now, in regard to parasite infections, I believe that when Lydian became a teenager, there were ancestral traumas in our family line that made us vulnerable to developing a severe parasite infection. We were estranged from both sets of our parents (Lydian’s grandparents) at that time in our lives as well. We didn’t know it at the time, but we had traveled to some areas of the world (Nepal and India) that were very close to where Lydian’s future husband was living in Myanmar. In Nepal, we got rabies vaccines (later, though these vaccines may have made us “sick”, I was very grateful to have had mine because a few years later, I was bitten by a rabid dog in Myanmar).
A lot of readers will focus in on the India and Nepal travel as the cause of our parasites, but while I can’t deny that there was exposure in India and Nepal on that trip, we returned to India and stayed there for several months after we overcame that heavy worm load from our first trip. We had no issues with parasites after this second trip, which was much longer, and that involved travel to much more challenging areas of India. So while exposure to parasites definitely can play a role in the development of a heavy worm load, my own personal experience with parasites is that exposure is not the whole story.
Click here to subscribe to the Living Database!
Life Stages in Helminth Life Cycle
Migration and Development: Once inside the body, larvae don't simply stay put. A well-documented pattern involves larvae traveling through the bloodstream to the lungs, from there moving up into the airway, and then being swallowed back down into the small intestine. Larvae take a circuitous route that has clinical consequences in terms of respiratory symptoms that can precede or accompany intestinal disease.Maturation and Reproduction: Larvae that complete their migratory phase settle in target organs, most commonly the intestines, where they develop into adult worms and begin reproducing. The reproductive output is substantial. A single adult female can produce thousands of eggs, which is part of what makes reinfection so persistent in some settings.
This is a disconcerting fact, of course, but it’s important to stay emotionally grounded if you’re doing self-treatment for parasites for yourself or your family. Parasites are everywhere and normally, our bodies resist infection if we have adequate levels of nutrients like iodine and potassium iodide and amygdalin.
Release into the Environment: Eggs exit the human body through feces and enter the external environment. From there, what happens next depends entirely on the species.
It’s important to note here that environmental release of eggs is a consideration in terms of treatment of parasites in humans. Many of the eggs are vulnerable to high heat. For the first 3 months of treatment, we therefore recommend that patients do steam-cleaning of floors and fabrics anywhere in the home where they (or their partner or their children) disrobe and remove their underwear. Eggs can be shed into the underwear and onto the floor and on sheets and fabrics. Eggs that end up on our feet or in underwear or on sheets can end up back inside a person’s mouth at night during sleep. Steam cleaning these areas once a month can help reduce the worm load by reducing the number of eggs that we’re exposed to through shedding and inadvertent re-infection that happens via the hands during sleep.
Clothing should be dried in a dryer on high-heat for the first 3 months of treatment in order to kill helminth eggs.
Development Outside the Host: Soil-transmitted helminths require time in the soil to mature before their eggs or larvae become infectious to another person. Other helminths depend on intermediate hosts. Snails, for example, are an intermediate host in the case of liver flukes. Cattle or pigs are intermediate hosts in the case of tapeworms. Intermediate hosts are needed in order to complete stages of development that the human host alone cannot support.
Diatomaceous earth can be mixed with soil that can be used to reduce the number of larvae in an area that’s known to be a source of helminth infection for humans or animals.
Two Main Types of Helminth Life Cycles
Direct Cycle (Soil-Transmitted): In the direct cycle, the parasite requires only a single human host to complete its life cycle, but it does need a short period of time in the external environment first. Eggs or larvae mature in the soil before becoming capable of infecting another person. Roundworms and hookworms are the classic examples of this pattern.Note that though the Direct Cycle is presented as “Soil Transmitted”, in fact, eggs can be shed onto tile or carpet inside the house and then re-infect the host. Soil is not required. But as noted above, steam-cleaning around areas where you walk naked in your house and drying clothes can help reduce this problem and make it a non-issue over the course of time. Do steam-cleaning once monthly. Dry clothes on high-heat for all laundry during the first 3 months of treatment.
Indirect Cycle (Vector or Intermediate Host): In the indirect cycle, the parasite cannot complete its life cycle without passing through one or more intermediate hosts. Schistosoma, for example, requires freshwater snails as an intermediate host. Taenia tapeworms require cattle or pigs as intermediate hosts. The intermediate host is a biological requirement in order for the helminth to survive, which is why public health measures targeting intermediate hosts (livestock management, snail control) can be effective levers for disease reduction.
The Alive-N-Healthy DMSO Guidebook - Buy Here!
The Life Cycle of Common Helminths That Infect Humans
Ascaris lumbricoides / Roundworms: Ingested eggs hatch in the small intestine. Larvae migrate to the lungs, where they can cause pulmonary symptoms, before traveling back up the airway and being swallowed to return to the intestine, where they mature into adults.Ancylostoma duodenale / Necator americanus / Hookworms Infective larvae in contaminated soil penetrate bare skin, most often the feet. They migrate to the lungs via the bloodstream and eventually mature in the small intestine, where adult worms feed on intestinal epithelium and blood.
Taenia solium / Pork Tapeworm: Humans ingest larvae encysted in undercooked pork. The larvae evaginate in the intestine and grow into adult tapeworms. If humans inadvertently ingest T. solium eggs rather than larvae, the result is cysticercosis, a considerably more dangerous disease in which larval cysts develop in the tissues, including the brain.
Schistosoma (Blood Fluke) Cercariae released from freshwater snails penetrate human skin during water contact. They migrate to the liver, mature, and eventually settle in the mesenteric or vesical veins, where adult worms deposit eggs. Trapped eggs drive the chronic granulomatous inflammation that underlies the most serious clinical consequences of schistosomiasis.
Symptoms of Helminth Infection
As we’ve already noted elsewhere, helminth “infection” is a bit of a misnomer. Rather, “heavy worm load” is a more apt phrase because a balanced human gut ecology contains a few helminths that balance immune system reactivity.One of the defining features of helminthic disease is its relationship to worm burden. Light infections, which are actually the most common presentation globally, frequently produce no symptoms at all. In fact, not having any intestinal worms can lead to crippling allergy symptoms. So having too few or too many intestinal worms is the problem. Nonetheless, we focus in this discussion on the heavy, chronic infestations that generate the clinical picture most people associate with parasitic worm infections.
Gastrointestinal effects are the most common presentation of heavy helminth infection and they include abdominal pain, diarrhea, nausea, vomiting, gas, and bloating. These symptoms reflect the direct mechanical and inflammatory effects of worms attached to or migrating through the gut. Many people also experience “anorexia” or a lack of appetite.
Intense itching is a common symptom that can be particularly severe as localized perianal pruritus that worsens at night. This is the hallmark symptom of a heavy pinworm “load”. This nocturnal pattern is directly tied to the female worm's egg-laying behavior and is often the symptom that brings the infection to clinical attention, especially in children.
Systemic and nutritional effects accumulate over time with chronic, heavy worm load. Weight loss, malnutrition, iron deficiency, and anemia are all documented consequences. In children, the stakes are higher. Chronic worm burden can impair physical growth and blunt cognitive development, compounding educational and economic disadvantage in ways that persist long after the infection itself is treated.
Skin manifestations can include a red, worm-shaped or serpiginous rash. This is most characteristically associated with hookworm larval migration through the skin along with more generalized rash, weakness, dizziness, and fatigue.
Severe complications, while less common, are clinically important. Very high-intensity infections can cause intestinal obstruction, rectal prolapse, and — in immunocompromised individuals — fatal hyperinfection, most notably with Strongyloides stercoralis, whose capacity for internal autoinfection allows worm burden to escalate rapidly when immune control is lost.
Anemia is caused by hookworm infections and a lack of adequate numbers of red blood cells can cause serious symptoms of brain fog, exhaustion, depression, chronic pain, and a wide array of other symptoms. Do not give iron supplements for anemia. Rather, we’ll talk about how to supply the body with copper as an essential nutrient for iron-deficiency anemia. DO take vitamin B12 supplements or administer vitamin B12 powder topically by combining it with dimethylsulfoxide (DMSO).

Parasites: Self-Treatment and Prevention - BUY THE eBOOK HERE!!!
Species-Specific Symptoms
While the symptom categories above apply broadly, individual helminth species produce recognizable symptoms that can be helpful for those who are self-treating at home:- Pinworms (Enterobius vermicularis): The defining symptom for pinworms is severe, localized anal itching, characteristically worst at night. In children, ADHD symptoms are common as a result of a lack of sleep.
- Hookworms (Necator americanus, Ancylostoma duodenale): Produce a distinct red, crawling rash at the site of larval skin penetration, and are among the most significant helminthic causes of iron deficiency anemia through chronic intestinal blood loss.
- Whipworms (Trichuris trichiura): Associated with chronic abdominal discomfort and diarrhea, and with heavy-burden complications including rectal prolapse in children. Rectal prolapse can be healed using herbal medicines.
Helminth Immunomodulation
Intestinal parasites are, in a very real sense, intimately tied to human existence. These organisms have been with us for as long as we've been on earth. Intestinal helminths are multicellular organisms belonging to two distinct groups, the nematodes and the flatworms, and they invade a range of organs (lungs, intestine, liver) through a variety of routes depending on the species. The downstream consequences of helminth infection in humans typically include malnutrition, growth disturbances, and cognitive impairment, particularly in children, and these negative results of infection tend to take center-stage. But what deserves closer attention here is something less commonly discussed, namely the fact that helminths can provide an important benefit to the human immune system, and how that works.Survival Through Immunomodulation
Helminths are long-lived parasites that do not replicate within the human host. That single biological constraintm, no internal replication, shapes their entire survival strategy. They can’t overwhelm the host through sheer numbers the way a rapidly dividing pathogen such as a bacteria or a virus can. Instead, they have evolved a sophisticated capacity for immunomodulation: the ability to reshape the host's immune environment in ways that prevent their own elimination while also protecting the host from the worst consequences of uncontrolled inflammatory response. This mutual benefit, reduced immuno-reactivity by the host, and the continued survival for the parasite, is thought to be the organizing logic of the helminth-host relationship.How Helminths Modulate Immunity
The immunomodulatory effects of helminths operate on multiple levels simultaneously.At the level of antigen presentation, helminths are able to interfere with the maturation of dendritic cells, the sentinels of the adaptive immune system, thereby altering how antigens are presented and steering the immune response preferentially toward Th2 and Th3 pathways rather than the pro-inflammatory Th1 responses that would threaten the parasite's survival.
Of particular significance is the ability of helminths to induce CD4+CD25+Foxp3+ regulatory T cells (Tregs). Tregs play a central role in maintaining immune homeostasis and in suppressing autoimmunity. They are, in a sense, the immune system's internal braking system. The helminth-driven induction of Tregs does two things at once:
- Helminth induction of Tregs contributes to the longevity of the parasite in the infected host, and it helps explain why helminthic infection appears to offer protection against certain autoimmune diseases, including multiple sclerosis and inflammatory bowel disease, as well as cancer. This is an important thing to consider as you set forth to reduce your worm load. It sits at the heart of the hygiene hypothesis and of a growing body of research into therapeutic applications of helminth biology. We need parasites and parasites need us. The goal of treatment is always to balance the body’s internal ecology, not to obliterate a species completely.
- It's also worth noting that comparable immunomodulatory phenomena have been observed in infections with protozoan parasites, Schistosoma and Leishmania for example, suggesting that this capacity to reshape host immunity is not unique to helminths but reflects a broader survival strategy among long-established parasites that have co-evolved alongside the human immune system.
The Double-Edged Sword
None of this comes without a cost. The same immunomodulatory capacity that protects the host from inflammatory damage and the parasite from elimination also suppresses the immune system's ability to mount an effective response to vaccination. Both oral and parenteral immunizations show are less effective in helminth-infected individuals.How we think about this helminth-vaccine problem is politically driven. What are the stories that we’ve been told, as humans, in regard to vaccines? What are the stories that we’ve been told about parasites? If you believe, dear reader, that vaccines are bad, then this statement about helminths and lowered vaccine effectiveness will inevitably trigger a specific reaction and bullet-point reasoning about why that relationship exists. If, on the other hand, you believe that vaccines are good, you might be concerned about the implications of a heavy worm load according to a very different story.
Heavy metal exposure, of course, comes to mind for anyone who is familiar with the political view that vaccines are bad. Studies have demonstrated that heavy metals tend to accumulate in parasites
Persistent infections, those caused by viruses and by parasites that establish long-term residence in the body rather than being cleared by the immune system, are associated with a wide range of chronic diseases and a substantial burden of mortality. At the same time, heavy metals are ubiquitous environmental pollutants with well-documented immunosuppressive properties. The question is: does heavy metal exposure suppress the immune system in ways that increase a person's susceptibility to these persistent infections? And if so, how much of that increased susceptibility runs directly through immune function itself?
One study that used data from NHANES 1999–2016 (a large, nationally representative dataset that makes it possible to examine population-level exposure and infection patterns over nearly two decades) looked at associations between heavy metal exposure and six persistent infections:
- Cytomegalovirus (CMV),
- Epstein-Barr Virus (EBV),
- Hepatitis C Virus (HCV),
- Herpes Simplex Virus Type-1 (HSV-1),
- Toxoplasma gondii, and
- Toxocara spp. (Toxocara canis and Toxocara cati)
The findings in this study were consistent and identified positive associations between multiple individual heavy metals and increased risk of persistent infection across several of the pathogens examined.
The study found that combined heavy metal exposure was associated with meaningfully elevated odds of infection across five of the six pathogens studied:
- Cytomegalovirus: OR 1.58 (95% CI: 1.17–2.14)
- Hepatitis C virus: OR 2.94 (95% CI: 1.68–5.16)
- HSV-1: OR 1.25 (95% CI: 1.11–1.42)
- Toxoplasmosis gondii: OR 1.97 (95% CI: 1.41–2.76)
- Toxocara spp.: OR 1.76 (95% CI: 1.16–2.66)
Arsenic, cadmium, and lead are the heavy metals with the most pronounced independent contributions to infection risk in humans. Heavy metals produce an increase in systemic immune inflammation (a measure of overall immune status). This inflammation definitely seems to increase vulnerability to infection, including parasitic infection.
The picture that emerges here is straightforward in its broad strokes and worth stating plainly: heavy metal exposure, the kind that is pervasive in modern industrialized environments. appears to compromise the immune system's capacity to control and resist persistent infections. Immune system compromise is at least partly responsible for the elevated infection risk. Reducing heavy metal exposure, by this logic, is not only an environmental health priority but a potentially meaningful lever for reducing the burden of chronic viral and parasitic infections. Heavy metal chelation therapy can also play a vital role in promoting better health overall.
Protozoa / Sporozoa Life Cycles
Sporozoa / Apicomplexa, have a complex life cycle that involves multiple hosts. Sporozoa alternate between asexual reproduction and sexual reproduction to produce different life cycle stages including:- Infectious sporozoites
- Feeding Trophozoites
- Resistant cysts (oocysts)
The Life Cycle of Common Sporozoa / Protozoa That Infect Humans
In the developed world, helminths tend to produce the biggest amount of woe for people who develop parasite infections, but in third world countries in Africa and Asia, malaria is a huge problem that kills at least a million people every year, mostly children.
Toxoplasma: Toxoplasma gondii is a protozoa that has a very broad host range. It infects most species of warm-blooded animals, humans included — and is the causative agent of toxoplasmosis. Its global prevalence makes it one of the most common parasitic infections in the world, yet it remains largely invisible in the popular imagination, in part because the majority of infections in healthy individuals produce no symptoms at all.
The only known definitive hosts for T. gondii, meaning the only hosts in which the parasite undergoes sexual reproduction, are domestic cats and their wild relatives. Cats shed unsporulated oocysts in their feces, typically for only one to three weeks, but in large enough numbers to constitute a significant environmental reservoir. Those oocysts require one to five days to sporulate in the environment before becoming infective.
Intermediate hosts such as birds, rodents, and other warm-blooded animals, acquire infection by ingesting soil, water, or plant material contaminated with sporulated oocysts. Shortly after ingestion, oocysts transform into tachyzoites, the rapidly dividing form of the parasite. Tachyzoites migrate to neural and muscle tissue, where they develop into the slower-moving bradyzoites housed within tissue cysts, a form the parasite can maintain indefinitely within the host. Cats close the loop by consuming infected intermediate hosts harboring these tissue cysts, though they can also become infected directly through ingestion of sporulated oocysts.
Animals raised for human consumption, as well as wild game, can similarly acquire tissue cysts by ingesting oocysts in the environment which is why food handling and meat preparation practices are an important variable to consider in terms of human infection risk.
Humans can become infected with Toxoplasma gondii through any of several routes:
- Eating undercooked meat from animals harboring tissue cysts
- Consuming food or water contaminated with cat feces, or through direct contact with fecal-contaminated soil (including litter box changes)
- Blood transfusion or organ transplantation
- Transplacental transmission from mother to fetus
Once inside the human host, T. gondii forms tissue cysts most commonly in skeletal muscle, the myocardium, the brain, and the eyes. These cysts have the capacity to persist for the entire lifetime in their human host, a detail worth keeping in mind when considering the parasite's long-term clinical implications, particularly in the context of immunosuppression or pregnancy.
In immunocompetent individuals, acquired toxoplasmosis is typically asymptomatic. Roughly 10–20% of people with acute infection develop cervical lymphadenopathy and a flu-like illness, but the clinical course is generally benign and self-limited, resolving within a few weeks to months. Ocular involvement with visual loss is a rare but recognized complication. In other words, toxoplasmosis is one example of a very common parasitic “infection” that often produces no symptoms and no poor human health outcomes per se.
In immunodeficient patients, the picture changes significantly. Central nervous system disease is the most common presentation, though retinochoroiditis, pneumonitis, and other systemic involvement can also occur. In patients with AIDS, toxoplasmic encephalitis is the most common cause of intracerebral mass lesions and is thought to arise primarily from reactivation of chronic, previously latent toxoplasmosis infection. This highlights the idea that tissue cysts established during acute infection don't necessarily disappear. They can wait for the immune system to go into a compromised, weakened state. In patients on immunosuppressive therapy, disease may reflect either newly acquired infection or reactivation of latent infection that predates their immunosuppression.
Congenital toxoplasmosis deserves particular attention. It results from an acute primary infection acquired by the mother during pregnancy and is transmitted transplacentally to the fetus. The incidence and severity of congenital disease vary with the trimester of infection. Treatment of the mother during pregnancy can reduce both the rate of fetal infection and the severity of sequelae in affected infants which arguably makes prompt, accurate diagnosis in pregnant women a genuine priority.
Many infants who appear subclinically infected at birth will go on to develop signs or symptoms of congenital toxoplasmosis over time. Ocular toxoplasma infection, an important cause of retinochoroiditis in the United States, can result from either congenital infection or postnatal acquisition. In congenital cases, patients are often entirely asymptomatic until the second or third decade of life, when ocular lesions first manifest. The long latency between infection and clinical expression is one of the more striking features of this disease and one that contributes to its being underdiagnosed.
It’s noteworthy that cataracts can be caused by toxoplasmosis infection. The idea that this is a parasitic disease that can remain dormant until the immune system is weakened in some way deserves mention. In order to successfully treat toxoplasmosis-induced cataracts, it would be valuable to know that this is the underlying cause.
Malaria Parasites / Plasmodium spp.: In the exoerythrocytic stage - schizogony, human infection results from a bite from an Anopheles mosquito. Sporozoites are injected into the bloodstream. Within about 1 hour of infection, they enter the parenchymal cells in the liver and the first stage of development takes place. Asexual reproduction produces merozoites that rupture and leave the liver cells, enter the blood, and invade red blood cells. Merozoites don’t return from red blood cells to liver cells. Rather, merozoites reproduce in a species-characteristic manner such that batches of merozoites are released in 48 to 72 hour intervals (depending on the species). Hemoglobin and iron is “stolen” from red blood cells, leading to the development of anemia in patients.
Mosquitos then bite the infected patient during a blood meal and in the mosquito’s stomach, a microgametocyte produces 8 more microgamates through a process known as exflagellation. Macrogametocytes develop directly into macrogametes. Microgametes fertilize macrogametes to generate zygotes. Zygotes then become motile and elongated. Some develop into oocysts that grow, rupture, and release millions of sporozoites that end up in the mosquitos salivary glands to await a moment for inoculation into a human.
Dormant forms of Plasmodium vivax or Plasmodium ovale might persist as hypnozoites after the parasites disappear from the peripheral blood supply. This can lead to a recurrence of the infection which periodically re-emerges for up to 5 years. Plasmodium malariae infection can persist with periodic recurrence for 40 years or more.
But that being said, it should be noted here that malaria fever therapy, also known as Plasmodium Therapy, can be used to cure diseases like cancer. So while untreated malaria is definitely a serious issue, each of the parasitic diseases also seems to have a balancing force on human health. This is a complex topic, of course, but it deserves consideration because it demonstrates that these diseases have a function that can be steered toward human health if you step outside of the restrictive model of conventional medicine and look through a different lens.
Cyclospora cayetanensis: Cyclospora cayetanensis is a coccidian protozoan and, as far as current evidence indicates, the sole species responsible for all human cases of cyclosporiasis. No animal reservoir for C. cayetanensis has been identified — humans appear to be the only major host. Occasionally, cysts are recovered from animal feces, but this is thought to represent spurious passage following coprophagy rather than true animal infection.
One of the clinically significant features of Cyclospora and a detail that distinguishes it from the closely related coccidian parasite Cryptosporidium is that freshly passed oocysts are not immediately infective. Direct fecal-oral transmission can’t occur. The oocyst must first undergo sporulation in the external environment, a process that takes days to weeks at temperatures between 22°C and 32°C. During sporulation, the sporont divides into two sporocysts, each containing two elongate sporozoites. Only at this point does the oocyst become capable of causing infection.
Sporulated oocysts contaminate fresh produce and water, which serve as the primary vehicles of human infection. Once ingested, the oocysts excyst in the gastrointestinal tract, releasing sporozoites that invade the epithelial cells of the small intestine. Inside those cells, the parasite undergoes asexual multiplication, producing type I and type II meronts. Merozoites from type I meronts appear to remain in the asexual cycle, perpetuating intracellular replication. Merozoites from type II meronts take a different path, they undergo sexual development into macrogametocytes and microgametocytes when they invade a new host cell. Fertilization follows, the resulting zygote develops into an oocyst, and that oocyst is released from the host cell and shed in the stool, returning to the environment to begin the cycle again.
Several aspects of intracellular replication remain incompletely understood, and the precise mechanisms by which oocysts contaminate food and water supplies are still under active investigation.
Cyclosporiasis has been reported across many countries but is most prevalent in tropical and subtropical regions. In the United States, the majority of cases occur during the spring and summer months, a seasonal pattern consistent with the temperature-dependent sporulation requirements of the oocyst. Outbreaks have been identified nearly every year since the mid-1990s, linked most commonly to contaminated fresh produce.
After an average incubation period of approximately one week, symptomatic infection typically presents as watery diarrhea of varying severity. Additional manifestations can include the complications of dysentery, broader abdominal symptoms, and nonspecific systemic symptoms such as headache and low-grade fever. What makes Cyclospora clinically distinctive is its duration. Untreated infections typically persist for ten to twelve weeks and may follow a relapsing and remitting course, considerably longer than most acute infectious diarrheas, which tends to prompt diagnostic workup when the timeline is recognized. Associated weight loss can be substantial.
In individuals with HIV or other immunosuppressive conditions, both the duration of symptoms and the degree of weight loss are amplified. In disease-endemic regions, on the other hand, infections are frequently asymptomatic, a pattern consistent with the partial immune tolerance that develops in populations with repeated exposure.
Cryptosporidium Cryptosporidium is an apicomplexan protozoan with a wide host range and a lot of species diversity. Many species and genotypes are capable of infecting humans. Some are zoonotic (they’re transmitted from animal hosts to humans), others are non-zoonotic (these are host-adapted, with no evidence of animal-to-human transmission).
The two leading causes of human cryptosporidiosis are Cryptosporidium parvum (formerly C. parvum genotype II) and Cryptosporidium hominis (formerly C. parvum genotype I). A number of additional species including C. meleagridis, C. felis, C. canis, C. ubiquitum, C. cuniculus, C. viatorum, Chipmunk genotype I, the Cryptosporidium mink genotype, and C. muris, have also been documented in human infections, though they are far less common.
Cryptosporidium hominis predominantly infects humans and is generally considered anthroponotic, though sporadic reports of infection in animal hosts exist. The zoonotic subtype families of Cryptosporidium parvum most frequently implicated in human infections are closely associated with cattle, particularly calves.
Sporulated oocysts that each contain four sporozoites, are excreted by infected hosts primarily through feces, and possibly through respiratory secretions as well. Transmission occurs mainly through ingestion of fecally contaminated water (drinking water or recreational water) or food (raw milk being a well-documented vehicle), or through direct contact with infected animals or people. Inhalation is a possible but it is a less established route.
Once a suitable host ingests the oocysts, excystation occurs. At this point, the sporozoites are released and go on to parasitize the epithelial cells of the gastrointestinal tract and, in some cases, the respiratory tract also. Inside these cells, the parasite first undergoes asexual multiplication (schizogony or merogony), then it transitions to sexual multiplication (gametogony), producing male microgamonts and female macrogamonts. When microgametes rupture from the microgamont and fertilize the macrogamonts, oocysts develop and sporulate within the infected host.
Two distinct oocyst types are produced.
- Thick-walled oocysts are excreted into the environment and serve as the source of new infections.
- Thin-walled oocysts are not shed in stool but instead, they remain within the host and participate in an internal autoinfective cycle, a mechanism that helps explain the persistence and escalation of infection in immunocompromised individuals.
One critical feature that distinguishes Cryptosporidium from closely related parasites like Cyclospora is that oocysts are infectious immediately upon excretion, which enables direct and immediate fecal-oral transmission without any required environmental maturation period. Extracellular stages have been reported but their significance within the overall life cycle remains unclear.
Zoonotic and non-zoonotic Cryptosporidium species and genotypes are found worldwide. Outbreaks of cryptosporidiosis are reported regularly across multiple countries and continue to occur. In the United States, documented outbreak sources have included swimming pools, water playgrounds and recreational water venues, unpasteurized cider, juice and milk, direct animal contact, childcare settings, camps, and ill food handlers. The breadth of that list is itself informative. Cryptosporidium is chlorine resistance, low infectious dose, and immediate oocyst infectivity make it a pathogen well-suited to causing outbreaks wherever water or food safety controls have any gap.
The clinical picture with Cryptosporidium spans a wide range, and the host's immune status is the dominant variable in determining where on that spectrum a given patient lands.
The incubation period averages seven days, with a range of two to ten days. In immunocompetent individuals, infection typically presents as a self-limiting diarrheal illness that resolves within two to three weeks. Diarrhea may be accompanied by fever or fatigue, and the small intestine is the primary site of involvement.
In immunocompromised patients with a Cryptosporidium infection, the situation changes significantly. Severe, life-threatening malabsorption and wasting are possible, and the internal autoinfective cycle — driven by thin-walled oocysts that never leave the body — allows the infection to perpetuate and intensify without any external re-exposure. Extraintestinal cryptosporidiosis, while less common, has been documented in the pulmonary and biliary tracts and, rarely, in the pancreas — a reminder that this organism is not strictly confined to the gut when the immune system is unable to contain it.
Resources
Abu-Shakra, M. et al. (2015). Chapter 36 - Parasite Infection and Autoimmunity. Retrieved March 17, 2026 from https://www.sciencedirect.com/topics/agricultural-and-biological-sciences/helminth#:~:text=Heterophyidae%20are%20a%20family%20of,Santos%20and%20Howgate%2C%202011).
Merdaw, M. A. (n.d.). Protozoa (Sporozoa). Retrieved March 17, 2026 from https://copharm.uobaghdad.edu.iq/wp-content/uploads/sites/6/2019/10/mlecture.pdf
Sures, B. et al. (1999). Parasites as accumulation indicators of heavy metal pollution. Retrieved March 17, 2026 from https://pubmed.ncbi.nlm.nih.gov/10234173/
Golinska, Z. and Bany, J. (2000). Impact of heavy metals on the host-parasite system. Retrieved March 17, 2026 from https://pubmed.ncbi.nlm.nih.gov/16886325/
Zhang, H. et al. (2024). Association between heavy metals exposure and persistent infections: the mediating role of immune function. Retrieved March 17, 2026 from https://pubmed.ncbi.nlm.nih.gov/39104887/
CDC (n.d.). Cyclosporiasis. Retrieved March 17, 2026 from https://www.cdc.gov/dpdx/cyclosporiasis/index.html
CDC (n.d.). Cryptosporidium. Retrieved March 17, 2026 from https://www.cdc.gov/dpdx/cryptosporidiosis/index.html