The Glutamate System and Pain
 The glutamatergic system plays an important role in both the transmission of normal pain and the transition into chronic pain conditions and the sensitization of the body centrally to chronic pain. In conventional medicine, several methods have been proposed to control or reduce pain through treatment of different aspects of the glutamate system including:
The glutamatergic system plays an important role in both the transmission of normal pain and the transition into chronic pain conditions and the sensitization of the body centrally to chronic pain. In conventional medicine, several methods have been proposed to control or reduce pain through treatment of different aspects of the glutamate system including:
- Antagonism of ionotropic and Group I metabotropic glutamate receptors
- Agonism of Group II receptors
- Agonism of Group III receptors
- Modulation of transporter function
- Reduction of neuroimmune cytokines that affect glutamate
- Improvement of glutamate transmission through diet
- Improvement of GABA transmission through diet
- Use of electroacupuncture
- Exercise
Dietary interventions that positively impact glutamatergic transmission and the reduction of inflammation produce few or no side effects and, as a general rule, should be considered as an adjunct or supportive treatment for chronic pain whenever possible.

Click here to schedule a health coaching session with us.
The Glutamate System and Neuropathic Pain
Neuropathic pain is caused by a dysfunction in the somatosensory nervous system. The glutamate system is powerfully involved in neuropathic pain but chronic pain medications in conventional medicine rarely target the glutamate system. When there is excess glutamate around pain transmitting neurons, glutamate transporters (also known as excitatory amino acid transporters or EAATs) remove the excess glutamate to decrease the sensation of pain.NMDA Receptors in the Glutamate System
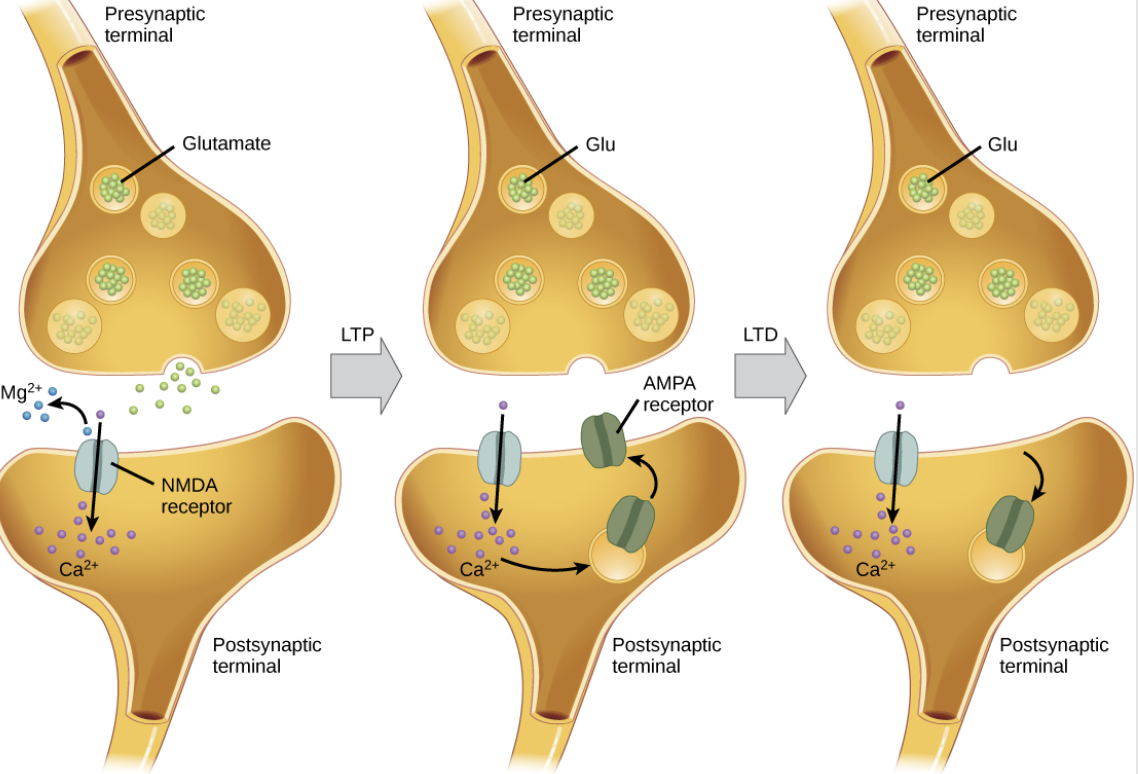
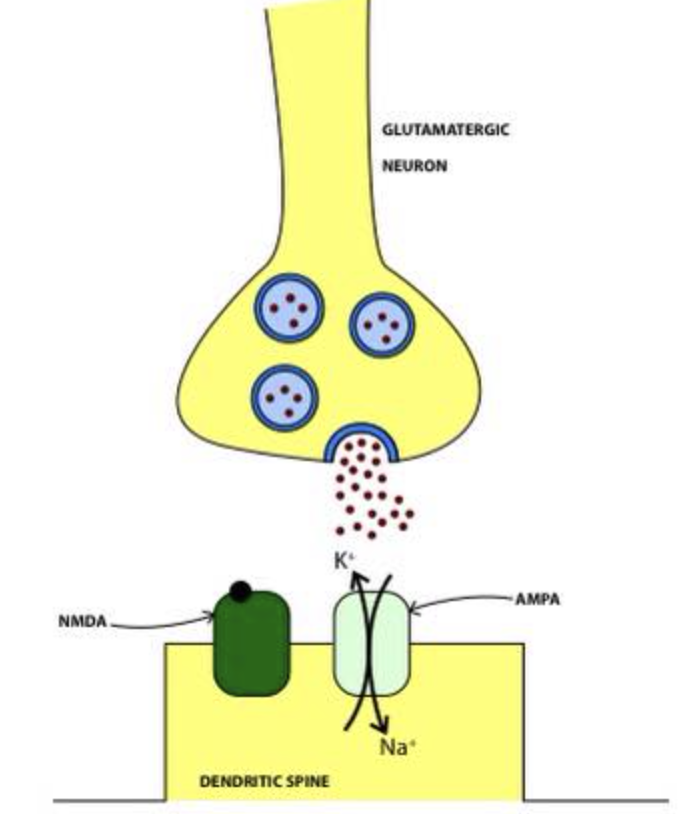
Glutamate is the primary excitatory neurotransmitter in the brain. It plays a role in synaptic plasticity or our ability to change our minds, adapt to, and overcome the environment and different social situations. The glutamatergic system is extremely important in the formation of new memories. Glutamate can bind to several different receptors including alpha-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptors, kainate receptors, and N-methyl-D-aspartate (NMDA) receptors.There are different subtypes of NMDA receptors. Each of the NMDA receptors has 2 N1 subunits and either two N2 subunits or two N3 subunits. The N1/N2 NMDA receptor complex is one of the most important subtypes in terms of treating chronic pain. The N1/N2 NMDA receptor complex precisely controls the cell membrane’s permeability to ions. When the proper permeability levels are disrupted via issues pertaining to the N1/N2 NMDA receptors, the cell may die or be seriously compromised in terms of its behavior. If many NMDA receptors are impacted, this can lead to chronic pain, memory issues such as the development of dementia or mental illness, or other serious neurological problems.

Become an Expert. Click here to do our online video courses.
To produce an electrical impulse in a healthy way, nerve cells that respond to glutamate must be able to open their ion channels and also close them. In glutamate-responsive nerve cells, ion channels open when certain things happen simultaneously to the cell.
- Two molecules of glycine or serine must bind to the NMDA receptor. While glycine can bind to extrasynaptic receptors, serine can bind to receptors that are located inside the synapse.
- Two molecules of glutamate must bind to the NMDA receptor.
- Note that even if #1 and #2 conditions are met, the ion channel may or may not open at this point.
- Magnesium or zinc can bind to sites on the NMDA receptor to block transmembrane ion channel opening. When this happens, calcium ions may not be able to enter the cell to produce a discharge. In order to produce a depolarization of the postsynaptic neuron, the magnesium and zinc must be dislodged from the cell.
- If an AMPA receptor is activated, it can depolarize the postsynaptic neuron and dislodge the magnesium or zinc, allowing the ion channel to open so that calcium can enter the cell.
NMDA receptors serve to notice and respond to specific neuronal coincidences. Ion channels in the cell membrane only open if the postsynaptic neuron depolarizes when glutamate enters the synapse. By reacting to coincidental events, NMDA receptors can have a grade response to stimuli. As such, there are three possible NMDA receptor responses that can occur:
- Short-term potentiation
- Long-term potentiation
- Excitotoxicity
Click here to subscribe to the Living Database now.
If there is only a small depolarization of the postsynaptic neuron, magnesium and zinc will only be partially dislodged to allow a small number of calcium ions to enter the cell. The calcium ions act as second messengers in this case, that temporarily recruit more AMPA receptors to the postsynaptic neuron. The number, activity, and position of AMPA receptors are dynamically changing constantly so that glutamatergic nerves can adjust their strength and activity to external needs and conditions. When more AMPA receptors are recruited to the postsynaptic neuron, this increases the chance for future depolarizations. This AMPA-recruitment effect is short-lived though. It only lasts for a few hours. As such, it is known as “short-term potentiation”.
A larger depolarization of the postsynaptic neuron can completely dislodge magnesium and zinc to allow a larger volume of calcium ions to enter a cell. The calcium ions then interact with genetic transcription factors to encourage the overall growth of the neuron. When a neuron grows and becomes larger, this is known as “long-term potentiation”. This is the mechanism behind synaptic plasticity and its effects can last for years or even for a lifetime. Neuron growth is the secret behind the brain’s ability to rewire itself but if the postsynaptic neuron depolarization is too prolonged and an unregulated amount of calcium ions enters the neuron, the neuron can die. This effect is known as “excitotoxicity”. When a postsynaptic neuron is overly depolarized to the point of death, this process can eventually lead to a variety of nervous system diseases and disorders, including chronic pain conditions.
NMDA receptors are nearly ubiquitous throughout the entire central nervous system and also within the peripheral nervous system. They are found on about 80% of the cells of the brain cortex and also within the hippocampus with preferential expression on the pyramidal neurons (which allow humans to take complex synaptic input and derive coherent output). NMDA receptors are also found on astrocytes and glial cells that have been traditionally viewed as neuron-support cells. They are found in the kidneys and in the heart in rats, though scientists have not been able to determine their function in these organ tissues.

Click here to do a free trial of EMDR to Release Trauma.
NMDA Receptor-Involvement in Excitotoxicity
The NMDA receptors belong to the glutamate system. NMDA receptors sometimes come into contact with excitotoxic-substances that cause over-excitement and over-stimulation of these receptors. Monosodium glutamate / MSG, domoic acid (found in shellfish), and L-beta-oxalyl-amino-alanine (BOAA), a glutamate analog, are food-based over-stimulators for the NMDA receptors that can cause neuron death and the development of serious neurological diseases including chronic pain conditions.
Over-stimulation of the NMDA receptors by toxins like MSG (and others) have been implicated in the development of the following diseases and disorders:
- Epilepsy
- NMDA receptor hyperexcitability plays a role in epileptic seizures. Uncontrolled neuronal firing involves unregulated calcium-ion influx and excitability which can cause brain damage.
- Stroke
- NMDA receptor hyperexcitability plays a role in brain damage caused by stroke. Uncontrolled neuronal firing after a stroke occurs involves unregulated calcium-ion influx and excitability which can cause additional brain damage.
- Traumatic Brain Injury (TBI)
- NMDA receptor hyperexcitability plays a role in traumatic brain injury. Uncontrolled neuronal firing following the injury involves unregulated calcium-ion influx and excitability which can cause additional brain damage.
- Alzheimer’s disease
- Memantine, an uncompetitive NMDA receptor antagonist improves Alzheimer’s symptoms, demonstrating the important role of the glutamate system in this disease.
- Migraine headaches
- Magnesium can be used to reduce the frequency and severity of migraine headaches, suggesting that NMDA receptors play an important role.
- Huntington disease
- Huntington disease involves NMDA receptor-mediated excitotoxicity. Amantadine and memantine are NMDA receptor antagonists that reduce the hyperexcitability of NMDA receptors to reduce symptoms of Huntington disease.
- Chronic pain conditions
- Major depressive disorder
- NMDA receptor antagonists like ketamine have shown great promise in the treatment of major depressive disorder.
- Tinnitus
- The NMDA receptors play a role in the development of tinnitus.
- Anti-NMDA Receptor Encephalitis
- Brain inflammation in Anti-NMDA Receptor Encephalitis is said to occur as a result of autoimmune attack on the NMDA receptors. We do not ascribe to a purist view of autoimmune disease because there’s so much scientific evidence linking autoimmunity to low-level infection or “colonization” by pathogens in the body that can be killed to restore health in those with autoimmune disease. Anti-NMDA Receptor Encephalitis is often associated with ovarian teratomas.
- Autism / ASD
- Autism / ASD is strongly associated with mercury exposure and mercury is an excitotoxic agent for the glutamate system.
- Heavy metal poisoning
- Lead is a heavy metal that can bind to NMDA receptors to produce certain toxic effects in terms of nervous system function.

Click here to learn more about the DreamLight.app, an online guided meditation and brain-entrainment tool.
Ketamine for Reducing NMDA Excitotoxicity
Ketamine was first produced in 1962 and in the 1970s, it was used as a relatively safe and effective anesthetic during surgery. Ketamine was used as a safer and more effective alternative to phencyclidine (PCP). Ketamine, in fact, was derived from PCP, but it had a much higher safety profile and produced fewer serious side effects as an anesthetic agent.
Of all of the NMDA receptor antagonists that have been scientifically studied for the treatment of neuropathic pain specifically, intravenous ketamine is the most effective. At least 13 clinical trials have found that ketamine is a powerful glutamatergic pain control agent for neuropathic pain. Like phencyclidine or PCP, another drug of abuse, ketamine interacts with NMDA receptors to reduce hyperexcitability.
Studies have shown that a variety of neuropathic pathologies respond to treatment with ketamine including:
- Spinal cord injury neuropathy
- Phantom limb pain
- Complex regional pain syndrome (CRPS)
Though ketamine therapy can significantly improve the quality of life for people with neuropathic pain conditions, it is not FDA approved at the time of this writing. This is no reason for people with chronic pain conditions to avoid ketamine though. The FDA, after all, exists primarily to support Big Pharma’s interests, not patient-interests.
Ketamine powerfully inhibits neuron hyperexcitability by binding noncompetitively to the phencyclidine site of NMDA receptors. In other words, ketamine acts as an NMDA receptor antagonist to dislodge over-stimulating substances that may play a role in causing chronic pain conditions. The S(+) enantiomer of ketamine (known as “esketamine”) has the highest therapeutic index and potency and the best side effect profile. When ketamine is administered, it is processed in the liver to an active metabolite (nor-ketamine) that is also analgesic.
 Everything You Never Wanted to Know About the Treatment of Chronic Pain... and How to Avoid Painkillers That Will Eventually Kill You - BUY HERE!!!
Everything You Never Wanted to Know About the Treatment of Chronic Pain... and How to Avoid Painkillers That Will Eventually Kill You - BUY HERE!!!
Low-Dose Ketamine and Intranasal Esketamine for Chronic Pain
Studies have shown that ketamine, when given as an intravenous infusion in low, sub-anesthetic doses, provides pain relief in nearly all instances and types of chronic pain with side effects that are typically mild. In some circles, we might call this “microdosing ketamine” for chronic pain relief, but in conventional medicine, low-dose ketamine is administered under a doctor’s care at a specific dose that does not cause psychoactive effects.Ketamine acts as an NMDA antagonist and it has been used as an anesthetic since the 1970s. Low-dose ketamine has more recently been studied in the treatment of phantom limb pain, postherpetic neuralgia, neuropathic cancer pain, and spinal cord injury. It is also used for pain relief after surgical procedures, fibromyalgia, and various other chronic pain conditions.
“Ketamine Clinics” have developed in recent years to treat chronic pain and depression in various countries throughout the world. These clinics exist in Mexico, the United States, France, South Korea, and the UK. Ketamine is legally administered in these clinics “off-label”.
NMDA receptors play an important role in chronic pain as well as mood regulation and the development of opioid tolerance. Both animal and human studies have shown that NMDA receptors play a vital role in the transition from acute, short-term pain to chronic pain conditions. The NMDA receptors play a key role in a reduction in pain tolerance and an increase in pain sensitivity.
Ketamine is an uncompetitive NMDA receptor antagonist that blocks the flow of calcium ions through the NMDA receptor by waiting until the receptor has been activated before binding to it. In other words, ketamine doesn’t bind to the same site that glycine and glutamate normally bind to. Ketamine and magnesium, for example, both inhibit NMDA receptor activity. Both magnesium and ketamine reduce the hyperexcitability of NMDA receptors by acting as “channel blockers”.
The pain-relieving effects of ketamine may be the result of NMDA receptor antagonism, but ketamine actually interacts with a number of nervous system receptors in the body including:
- Opioid receptors (mu, kappa, and sigma)
- Monaminergic receptors
- Dopamine receptors
- Serotonin receptors
- Cholinergic receptors
- Muscarinic receptors
- Glutamate receptors
- GABA receptors
This impressive list of receptors that ketamine can interact with can explain to some extent why it works so well to reduce chronic pain and alcohol addiction, opiate addiction, and major depression. The NMDA receptor interactions with ketamine are just one facet of how this medication works to treat chronic pain conditions.
Low Dose Ketamine - Dosing:
Low-dose ketamine is administered at a dose of 0.5 to 1 mg / kg to produce an effect that is less than what’s needed to cause a loss of consciousness or deep anesthesia.
The highest bioavailability of low-dose ketamine is through IV, but other methods of administration can also be used. The highest bioavailability of ketamine administration in descending order is:
- Intramuscular (about 93% bioavailability)
- Intranasal (about 45%)
- Sublingual (about 24-30%)
- Oral (about 17-24%)
About 80% of ketamine is first metabolized in the liver to norketamine. Norketamine has about a third of the pain-relieving effects of ketamine, but the norketamine half-life is longer. The total elimination half-life of ketamine is about 6 hours, but despite this, many patients have effects from ketamine treatment that can last for several months. Scientists believe that ketamine treatment can improve the emotional aspects involving pain. Indeed, ketamine in full-psychedelic doses often produces powerful visions and spiritual experiences that defy scientific inquiry. Nonetheless, it has been established that ketamine works through an impact on NMDA receptors but also a variety of other nervous system receptors and emotional-spiritual aspects of the human experience to produce pain-relieving results.
Ketamine is one of several important treatments that cure PTSD and depression and this medicine can have a lasting impact on chronic pain. Multiple treatments are often required. People who have chronic pain daily, after all, can experience “wind-up” pain in anticipation of the actual chronic pain, making the overall experience of physical pain much worse. This “wind-up” of anticipatory pain can maintain the chronic pain condition which may require a type of treatment like ketamine that can halt it and provides the patient with a sense of control.
Kratom / Mitragyna speciosa
Studies have shown that the mitragynine in kratom has the ability to diminish the excitability of NMDA receptors. Mitragynine by itself (not the whole plant), mimics the effects of AMPA and NMDA receptor antagonists in situations involving the long-term potentiation of neurons, but the whole herb performs differently than pure mitragynine, of course. Nonetheless, studies have shown that kratom has an impact on the glutamatergic aspects of chronic pain in addition to its ability to interact with opioid receptors for pain relief without itself acting with the same addictive potential as a true opiate.Kratom has an impact on arachidonic acid (the precursor to prostaglandins), pantothenate, coenzyme A, and tryptophan pathways in the human body to produce a powerful and reliable pain-relieving effect, but its impact on opioid receptors and NMDA receptors are also noteworthy. This herb is highly politicized because it poses a threat to Big Pharma and the opioid-addiction industry. Funding for research is geared toward the production of propaganda against responsible use as this is one of the most important herbs that people can work with to overcome opioid addiction especially when chronic physical pain is an underlying issue.
Like methadone, kratom interacts with the mu-opioid receptors while acting at the same time as an NMDA antagonist to treat and help people overcome opioid addiction except without the same level of risk that methadone carries of severe side effects and addiction.
Kratom is a fascinating herbal medicine that offers the most reliable pain-relieving effects in whole-plant form. Like ketamine, kratom as a whole herb, interacts with a number of different receptors in the nervous system, including dopamine and serotonin receptors as well as opiate receptors and NMDA receptors. Kratom can be administered in capsules as the dried herb at a dose of 500 mg to 6 grams daily to produce significant pain-relieving effects. Administer one particular “vein” (red, white, green, or yellow / gold) one day and another vein the next to reduce the odds of becoming even mildly addicted to kratom. Note that Red Hulu and Red Bali are some of the best strains for sleep.
Piracetam
Piracetam is a nootropic supplement that acts to produce cognitive enhancement through the NMDA receptors. Piracetam, in particular, when taken in fairly high doses (12,000 mg by mouth daily), can reduce anticipatory pain in part by improving the cellular membrane structure to make cells more “rounded” and more “bouncy”. Piracetam must be administered long-term in order to increase NMDA receptor density while significantly decreasing the anticipatory pain response.Piracetam can be taken at a dose of 12 grams daily along with Mitragyna speciosa to reduce overall pain in the body. Both kratom and piracetam heal the body as they reduce pain. Be aware that this is a medicine that must be taken for up to 6 months religiously in order to experience the full healing results.
Click here to buy piracetam powder.
Resources: